🧬Sable Bio: Building the Safety Layer Drug Discovery Has Been Missing
Deep Dive | Edition 18
Welcome back to the deep dive, where we break down the AI tools and data reshaping how new drugs are discovered. In each edition, we speak directly with the teams behind these tools to explain what they solve, how they work and where they are going next.
Keeping up with AI x life science news can get exhausting.
It’s scattered across LinkedIn, X, Substack, arXiv, Slack, newsletters... and you still somehow miss the things that actually matter. Too much noise, not enough signal.
We’re building something to fix that: a smarter, more powerful way to stay on top of what’s actually relevant to you.
But we want to build it with you, not just for you. Take 2 minutes to tell us what’s missing. What you share will directly shape what we build, and you’ll be the first to benefit from it.
Around 30% of clinical trials fail due to safety concerns. That’s billions in sunk costs, years of lost time, and patients who don’t get the medicines they need. Sable Bio thinks the problem starts much earlier in the pipeline, with how safety assessment is done in the first place.
I spoke with Olly Oechsle, CTO of Sable Bio, about how time-consuming traditional safety workflows can be and how the company’s Target Intelligence platform helps toxicologists.
🔴 The Problem
Before a drug candidate moves into preclinical development, safety scientists need to answer a deceptively simple question: if we inhibit or activate this target, what else is going to happen that we didn’t intend?
Answering that means looking across clinical trial data, genetic association studies, mouse knockout phenotypes, scientific literature, expression profiles, and more. Each lives in a different database, requires different expertise to interpret, and few are built with a toxicologist’s specific needs in mind.
The result is a process that takes anywhere from a few days to a month. Safety scientists spend a lot of that time collating information rather than doing the analytical work they’re trained for: identifying risks, building mitigation strategies, and making judgment calls about whether a target’s safety profile balances its therapeutic potential.
There’s also a reproducibility problem. “If another scientist were to do the same research with the same amount of time, would they have come up with the same answer?” Olly asks. When you’re manually searching PubMed, reviewing mouse knockouts or sifting through clinical databases, you inevitably go deep on some rabbit holes while missing others entirely. There’s no systematic way to know whether every potential adverse event has been examined across every relevant data source.
And the challenge doesn’t end with a single assessment. Drug discovery programs run for years. New data emerges, new papers are published, and safety scientists, often stretched across multiple projects, need to stay on top of all of it.
💡 The Platform
Sable’s core product is Target Safety Reports. A user searches for any target in the genome, specifies whether they plan to inhibit or activate it, and receives a comprehensive, customized safety report.
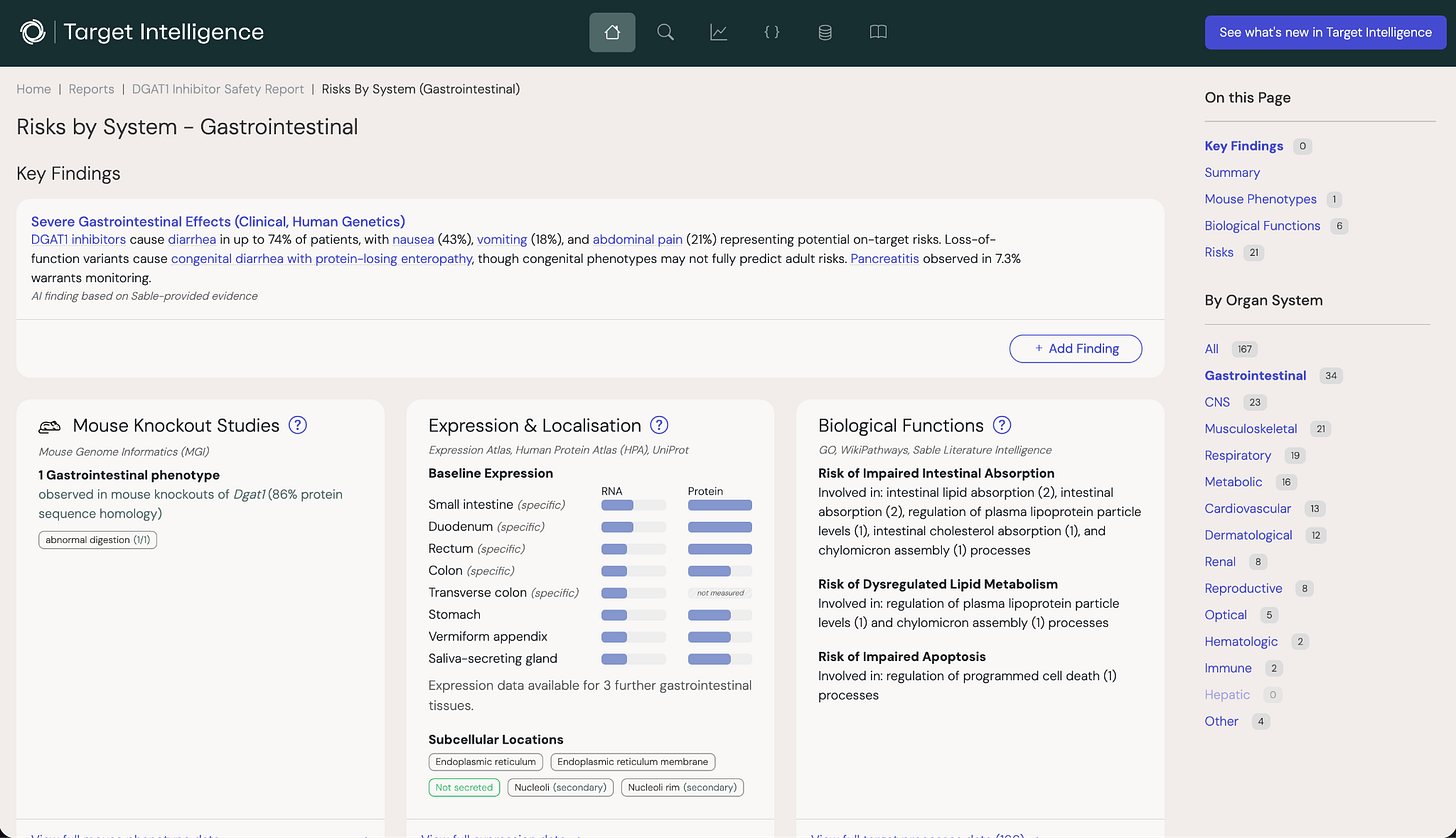
Findings can be organized by organ system (cardiovascular risk, hepatic risk, and so on) or by data source, letting users drill into the literature, clinical data, genetic associations, or expression profiles separately. The platform pulls from PubMed, clinical trial databases, GWAS data, gene burden studies, and MGI mouse knockout data, among other sources.
What makes this more than a search engine is how Sable has tuned each data source specifically for safety science and collated the result into a coherent report. For clinical trial data, statistical methods distinguish drug-centric effects from target-driven ones, and on-target from off-target effects, accounting for patient comorbidities that can be mistaken for causative drug effects. For the literature, Sable has built proprietary language models that extract target-to-adverse-event relationships with precision and coverage that general-purpose LLMs can’t match.
“It’s easy to make early strides with literature but it’s really hard to do it well,” Olly says. General AI models can answer questions about a single paper, but they struggle with corpus-wide analysis, and tend to “enthusiastically offer insights” from their broader training data that goes beyond the evidence being reviewed. Meanwhile literature tools can quickly present an overwhelming volume of content.
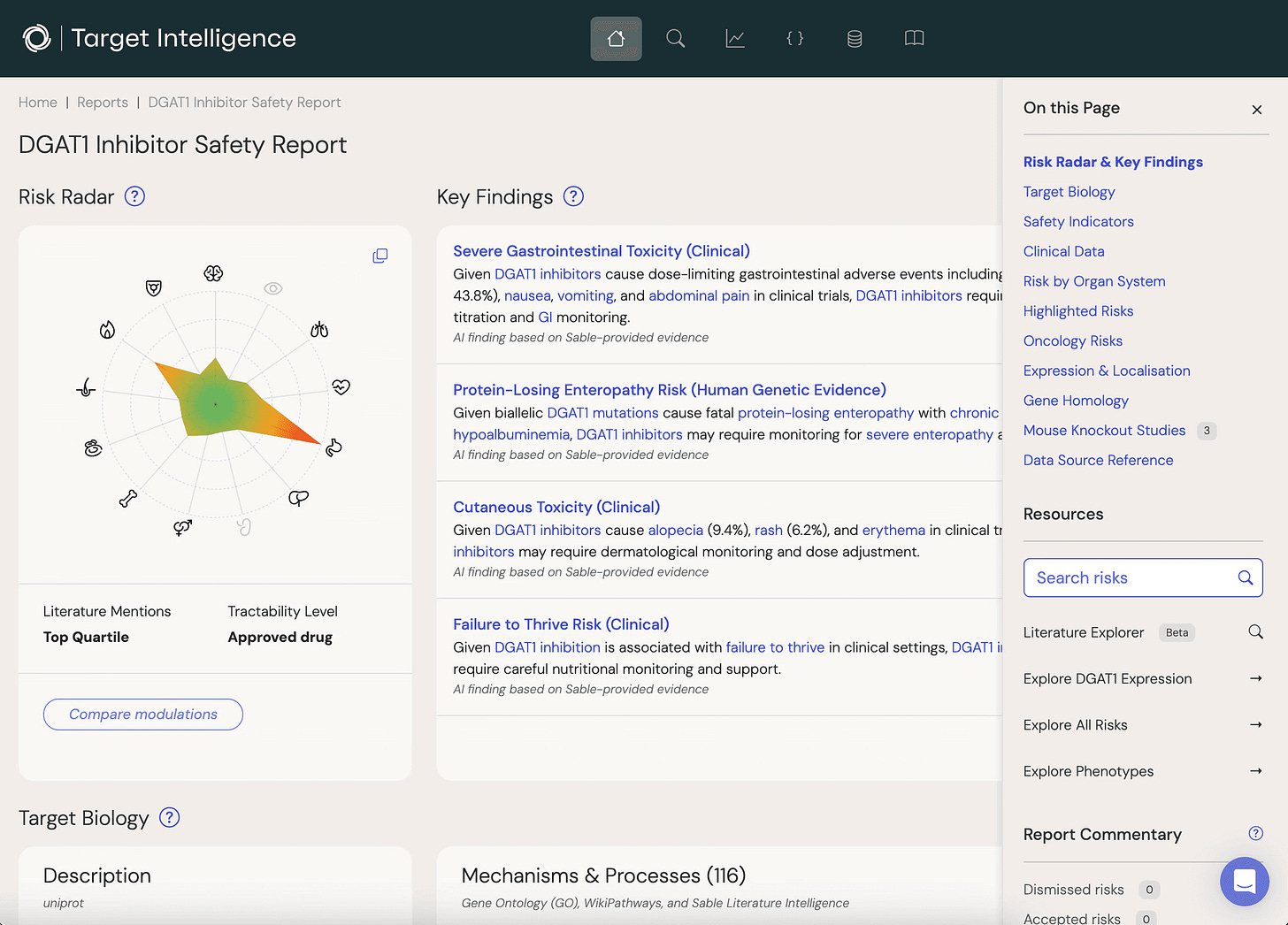
The platform also includes tracking that alerts users when new information emerges affecting a target’s safety profile, plus collaboration features for commenting, discussing, and sharing reports across teams.
📊 Weight of Evidence
Central to Sable’s approach is the toxicology principle of “weight of evidence.” Rarely does a single data source give a definitive answer on target safety. Instead, signals come from multiple directions: a suggestive mouse knockout phenotype, a mechanism described in the literature, expression data showing the target is active in a particular tissue, and adverse events observed with a related ligand.
Sable brings all of these signals together, letting scientists evaluate the full picture rather than chasing individual threads across separate databases. This works both ways: sometimes the different evidence types show a perceived hazard isn’t actually a concern, potentially saving organizations from expensive and unnecessary preclinical studies.
🧩 Where It Fits
Sable’s sweet spot is late discovery through to lead optimization, where the question shifts from “will this work?” to “what else is this doing?” The platform is used by preclinical safety scientists at several top-10 pharma companies, biotechs, venture capital firms conducting asset due diligence, and by consultants running target safety assessments.
The team is exploring expansion into earlier target selection, and for organizations using AI-driven target ID platforms that generate dozens or hundreds of candidates, Sable is developing wider-scale safety assessment products, systematically evaluating 50 to 200 targets with a traceable decision-making record. These are planned for release within the year.
Beyond the web interface, Sable offers API access and is building MCP integrations, positioning itself as a universal safety layer that plugs into the broader drug discovery ecosystem.
🔮 The Future
Sable is rolling out its new proprietary literature models alongside deeper analysis for single-cell data, expression data, and mechanistic biology. The company is also running a side-by-side comparison study between toxicologist assessments and platform outputs, with results expected in a couple of months.
“We’re not there to replace the decision-making process, which is ultimately a human one,” Olly says. “We’re there to save experts’ time.” Given how much of that time currently goes toward collecting data rather than acting on it, that’s a proposition most safety scientists can get behind.
🧑🔬Get in touch with Olly.
Thanks for reading Kiin Bio Weekly!
💬 Get involved
We’re always looking to grow our community. If you’d like to get involved, contribute ideas or share something you’re building, fill out this form or reach out to me directly.
Subscribe now to stay at the forefront of AI in Life Science and keep up with this upcoming season of deep dives.
Connect With Us
Have questions on this or suggestions for our next deep dive? We’d love to hear from you!
📧 Email Us | 📲 Follow on LinkedIn | 🌐 Visit Our Website



